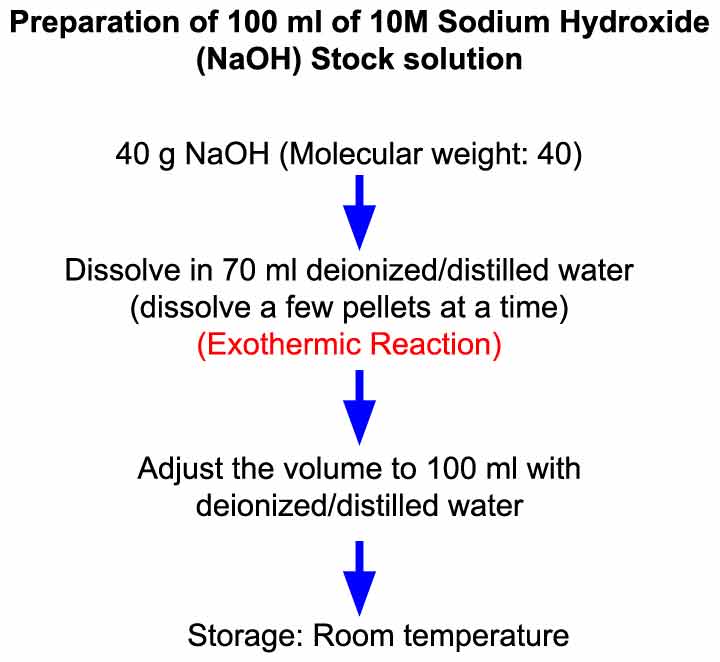
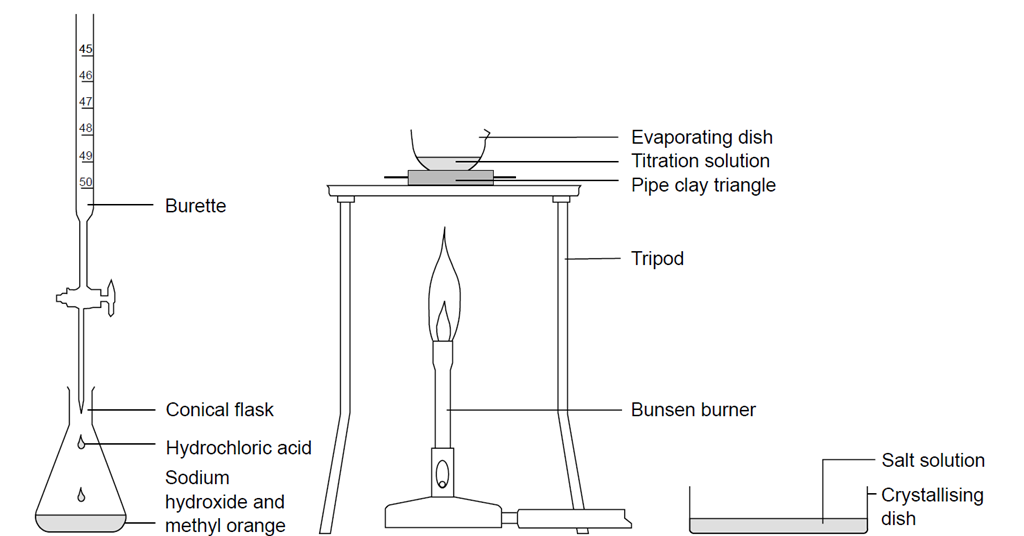
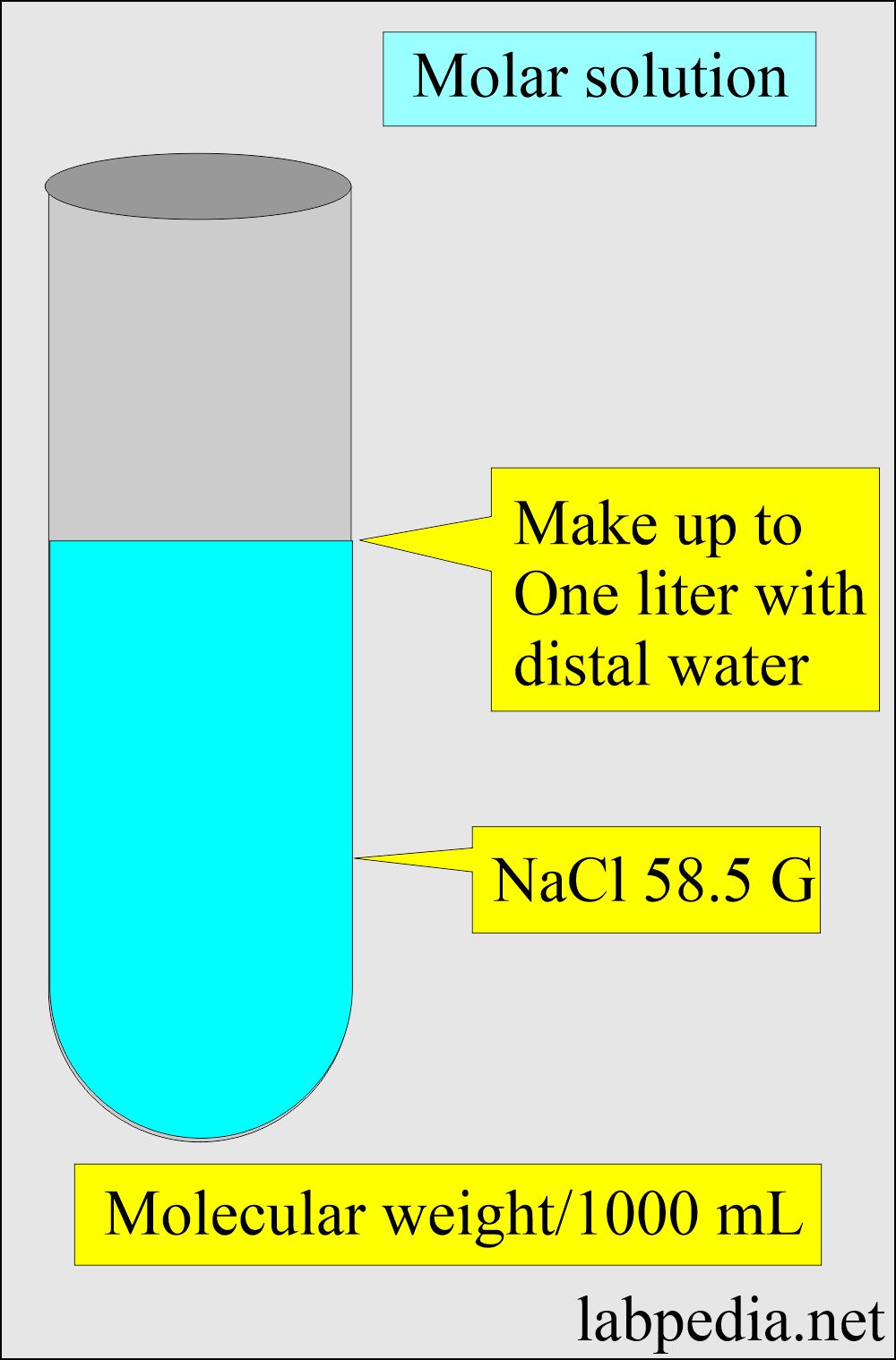
How to prepare N/10 NaOH solution - Chemistry - Some Basic Concepts of Chemistry - 7762175 | Meritnation.com

100mL` of `10% NaOH(w/V)` is added to `100 mL` of `10% HCI(w/V)`. The nature of resultant solut... - YouTube

Calculate the molarity of NaOH in the solution prepared by dissolving its 4 g in enough water to... - YouTube

SOLVED: Calculate the pH of a 6.7 X 10-2 M NaOH solution. ( a strong base) ( Your answer should have 2 digits after the decimal)

How to prepare 1% sodium hydroxide (NaOH), 5% NaOH, 10% NaOH solutions: Calculation and Explanation - YouTube

SOLVED: Aim: To determine the normality ofan unknown NaOH solution Solution in the burette is Normality of titrant (solution in the burette ) is Solution in the titrant flask is Volume of

Calculate molality of 2 molar NaOH solution which is 10% by weight - Chemistry - Solutions - 12944633 | Meritnation.com







:max_bytes(150000):strip_icc()/prepare-sodium-hydroxide-or-naoh-solution-608150_FINAL-696b52d6f90b4b1383ec8f95db73a1f3.png)